Coastal Field Guide
Guide Sections
- Introduction
- Preparation for Field Work
- Natural History
- Phenophase Definitions
- Terminology
- Data Sheets (printable)
- Data Entry Links
Rockweed Protocol
- Procedure A — Phenophase Observation
- Procedure B — Water Quality
- Procedure C — Age and Growth Assessment
The Signs of the Season’s Coastal Field Guide is adapted in part from Nature’s Notebook, USA National Phenology Network.
Introduction
This coastal species field guide is focused on observing the rockweed species, Ascophyllum nodosum, in the intertidal zone of the Maine and New Hampshire coast. There are three procedures to consider in monitoring Ascophyllum. Each is focused on a different aspect of rockweed research. All data will be entered using the links, which are found at the bottom of this page and in the Recording your Data section of the Resources for Coastal Observers page on the Signs of the Seasons website.
Procedure A: Phenophase
Participants will observe the phenophases of Ascophyllum by examining the presence and appearance of receptacles.
Procedure B: Water Quality Measurements
Two water quality parameters affecting the growth and phenophases of Ascophyllum are temperature and salinity. The water quality monitoring will be conducted each time the phenophase observations are made and also at the time of the age and growth assessment.
Procedure C: Age Assessment and Growth Rates
You may assist the research further by Assessing the Age and Growth Rates of the individuals you are observing in Procedure A. This should be accomplished once per year and typically in March/April, though it can be done at any time during the season.
Each of these Procedures is described in the instructions of this guide and training will be provided. Equipment, when available, will also be provided. We greatly appreciate your time, interest and assistance in conducting this research.
Preparation for Field Work
Before you go into the field:
- You will need to assess the tidal stage and plan accordingly. Tide charts may be found by selecting your region and location at Maine Boats and Harbors website where you can print monthly tide charts.
- Make certain you are prepared for the weather conditions, and that it is safe. Check the weather forecast for the time you will be in the field and wear appropriately safe shoes and clothing.
- Make sure you have the following field supplies and equipment: a transect line (soft tape measure), field hand lens, clipboard with field guide and data sheets, pencil, water quality equipment (thermometer and refractometer).
Natural History of Rockweed
Appearance
Ascophyllum nodosum, sometimes called knotted wrack, is a member of a brown algal (seaweed) group called “rockweeds”. It forms single bladders in long, strap-like fronds, which hang downwards, draping sheltered intertidal rocks. A number of fronds grow from each holdfast (where the seaweed is attached to the rock).
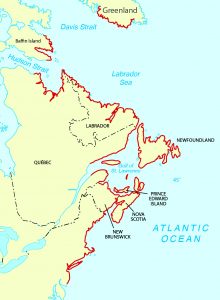
Range
Along the northeast Atlantic coastline, it ranges from NJ to Baffin Island, Canada. States where it is found: NJ, NY, CT, RI, MA, NH, ME.
Distribution
Found abundantly in the mid-intertidal zone on sheltered, hard substrate. A hardy organism, Ascophyllum nodosum can survive in varying environments and is tolerant of fluctuating temperature and salinity levels.
Role in the Environment
Rockweeds are conspicuous on North Atlantic rocky shores and provide habitat, shelter, and resources for a variety of marine biota.
Life Span
This species of rockweed is long-lived (a perennial plant): studies have documented that individuals can live for decades!
General Phenology and Life History
Each individual is either male or female. Reproduction appears to be correlated with temperature (Bacon & Vadas 1991)—the onset of reproduction generally occurs when water temperatures are 6˚C (43˚F), with peak reproduction at 10˚C (50˚F).
Similar to the way in which scientists use tree rings, the vesicles of Ascophyllum nodosum provide a record of its annual growth, showing which years had favorable or unfavorable conditions. Each spring, individuals create a new vesicle (airbladder) at the tip of each frond. Measuring the distance between vesicles provides a measurement of the growth rate from one growing season to the next.
Bacon, L.C. and R. L. Vadas (1991). A model for gamete release in Ascophyllum nodosum (Phaeophyta). Journal of Phycology 27 (2): 166-173
Rockweed Protocol
PROCEDURE A. Ascophyllum nodosum (Rockweed) Phenology Instructions
PLEASE BE CAREFUL! The intertidal zone is a slippery place. Your safety is our #1 concern. Please wear sturdy, closed-toed shoes, dress appropriately for the weather, and walk slowly on slick, uneven terrain. We highly recommend this monitoring be conducted in pairs for safety.
Please plan to evaluate the state of reproduction for Ascophyllum ideally one time per week (especially during the reproductive phases which start in very early spring) by observing the phenophases. If that is not feasible, make your observations twice a month. Pay particular attention to tide levels. In general, you will be able to access the shore one and a half hours before and after low tide. If your area is higher than high tide level, it may be accessible at all tide levels.
- For each monitoring month, establish a representative transect (approximately 10 m) that runs parallel to the water line, and passes through a patch of Ascophyllum nodosum in the intertidal zone. The location for your site should be mid-intertidal in the stand of rockweed. Choose a site where you can return to the same area each time.
- Haphazardly choose 10 individuals along the transect line. When handling individuals, take care. Although they are quite robust, they can be broken from their holdfast and will not reattach. It is difficult on some individuals to distinguish the primary shoot — in this case choose the longest frond close to the middle to check for phenophases. Avoid observing damaged fronds. There may be different phenophases occurring on one individual — in that case, establish the majority phase of the individual.
- For each individual, evaluate the state of the receptacles (see photographs) to determine what phase it is in. Do this for all 10 individuals. You do not need to count the receptacles. The value for each phenophase would be how many plants are in that phase out of the 10 plants. (Essentially, how many plants out of the ten are in that particular phase at the time of your observation? Write that number (value) after Y or N or ?). After recording the phenophase on the Phenophase Observations (A) Datasheet (PDF), enter your observations on-line at the SOS website on the SOS Coastal Observers page.
Note: This procedure should be conducted with water quality measurement for salinity and temperature (Procedure B).
Ascophyllum Phenophase Protocol adapted from GLOBE® 2005 Seaweed Reproductive Phenology Protocol, Earth System.
Phenophase Definitions for Observation
Directions: Record observations on a subset of 10 individual rockweeds at your site by determining how many observed individuals are in each phenophase. Do not make observations on rockweed individuals next to each other but every other one or haphazardly along the 10 meter transect line.
As you report on phenophase status (Y, N or ?) on the datasheets (refer to the definitions on this sheet to find out what you should look for) for each phenophase of this species. To report the intensity of the phenophase, choose the best answer to the question below the phenophase definition.
Do you see…
(Photos by GLOBE Project & E Stancioff)
PROCEDURE B. Water Quality Instructions
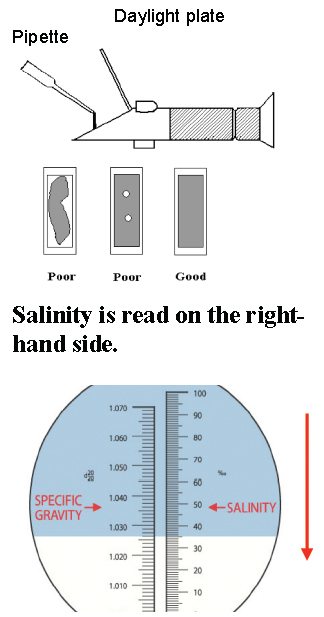 Instrument Preparation for Field Work: The refractometer should be calibrated each time observations are made, preferably before traveling to the site. First, open the lid and place several drops of distilled water on the daylight plate. Keeping the refractometer in a horizontal position, gently close the lid and be sure the sample is evenly distributed with no bubbles across the daylight plate. Look through the eyepiece and, if it appears blurry, turn the black ring to put the scale into focus. Loosen the calibration ring with the screwdriver and turn the ring until the boundary line between the white and blue regions crosses the right-hand scale at 0 (0/00). When this is complete, tighten the screw in the calibration ring. Use a Kim wipe to remove the distilled water. The refractometer is now ready for field use.
Instrument Preparation for Field Work: The refractometer should be calibrated each time observations are made, preferably before traveling to the site. First, open the lid and place several drops of distilled water on the daylight plate. Keeping the refractometer in a horizontal position, gently close the lid and be sure the sample is evenly distributed with no bubbles across the daylight plate. Look through the eyepiece and, if it appears blurry, turn the black ring to put the scale into focus. Loosen the calibration ring with the screwdriver and turn the ring until the boundary line between the white and blue regions crosses the right-hand scale at 0 (0/00). When this is complete, tighten the screw in the calibration ring. Use a Kim wipe to remove the distilled water. The refractometer is now ready for field use.
- Wade or reach into the water at the closest point to the rockweed transect that you are observing. Do not disturb the bottom. Take the temperature reading, and pipette the salinity sample at the exact same location ideally 5 to 7 inches below the surface of the water; note that often this can only be a few inches due to the tidal stage and safety. Submerge the thermometer probe until a steady reading is displayed. Submerge the pipette, rinse once and fill.
- In a stable spot (away from the water), record the water temperature on field data sheet and measure salinity using the hand-held refractometer.
- Place two drops of sample water from the pipette onto the daylight plate. Gently close the lid over the daylight plate. If necessary, point the refractometer towards or away from the sun to brighten or darken the field of view. You can also adjust for your eyesight by twisting the black ring near the eyepiece to focus the field of view. Read the salinity value on the right hand side. The value is where the boundary line of the blue and white fields cross the scale. Record the value on the field data sheet.
- Rinse the daylight plate with distilled water and wipe clean with a Kim wipe. (If the daylight plate is smeared with oil or similar liquids, it will repel the sample and alter the measurement).
- Write your data for water quality parameters in the Water Quality (B) data section of the datasheet and enter on the SOS website in the SOS Coastal Observers page.
Water Quality Methods adapted from: Stancioff, Esperanza (1992, revised 1996). Clean Water: A Manual for Coastal Water Quality Monitoring, published by Maine Sea Grant.
PROCEDURE C. Assessing Age and Growth Rate for Ascophyllum Instructions
Note: For both aging and growth rate, choose intact individuals and conduct this procedure once per year at any time during the season; though the preference is to conduct this in March/April.
- To obtain the minimum age of the plant, count all the vesicles (air bladders) on an intact primary shoot (if difficult to find primary shoot, select a shoot towards the middle of individual). Ascophyllum nodosum is dichotomous in branching, meaning the frond splits in a “Y”- follow one shoot all the way up. For example, in the line drawing of A. nodosum, the individual is at least 4 years old. Counting on the primary shoot, from the holdfast upwards, there are 4 vesicles (noted with yellow circles). Count and record the number of vesicles present, from the bottom to the top (oldest growth to the newest growth).
- To measure growth rate, measure the distance between each internode section (from mid-vesicle to mid-vesicle; letter “I” in the line drawing). This measurement is the growth in one year. Measurements of all the intermodal sections for an individual will allow one to see differences in growth rates between years and compare to other individuals. Measure growth rate from the newest internode to the oldest internode– so from the top down.
- Write your data for water quality parameters and growth rate on the Ascophyllum Age and Growth Rate (C) datasheet (PDF), and enter online on the SOS website at the SOS Coastal Observers page.
Note: This procedure should be conducted with water quality measurement for salinity and temperature (Procedure B).
Ascophyllum Growth and Aging Protocol modified by Jessica Muhlin, Maine Maritime Academy.
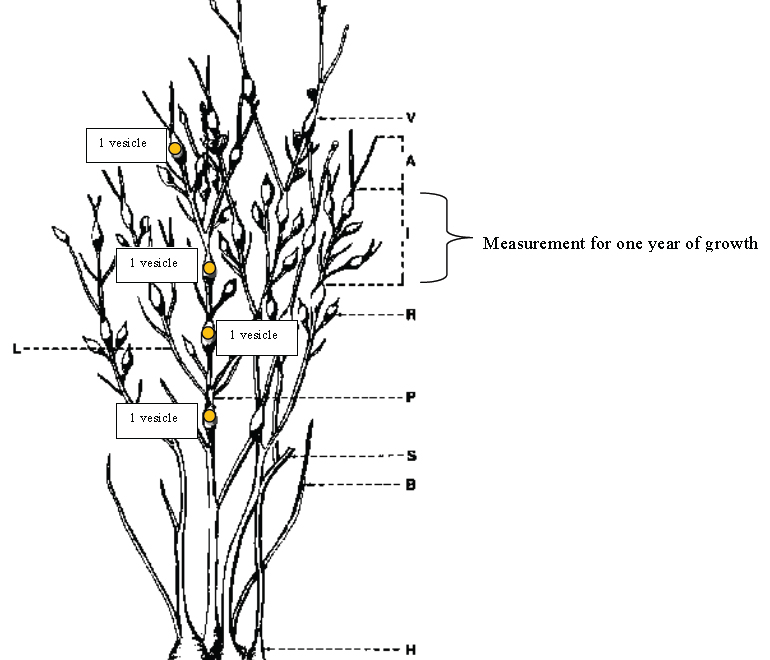
Terminology for morphology of Ascophyllum nodosum
Stump: When the body of the plant (called the thallus) is cut so that the branch of the alga is initially missing the region where growth occurs (the apical meristem).
Internode: The region of growth that is between two air bladders (vesicles).
Apical tip: The tips of the plant where growth occurs (they grow from the tips of the thallus).
Plant: an assemblage of fronds (branches) arising from a common holdfast.
Stand: a group of plants within a defined area.
Vesicles: Airbladders on that thallus. These are dilations of the shoot produced at intervals related to the rate of annual shoot elongation.
Receptacles: Reproductive organ of the plant. Receptacles are produced along lateral (side) shoots
Shoot: A part of the thallus resulting from growth. Shoots are analogous to branches of a tree.
Primary shoot: The main branch (trunk) of the individual, originating from the holdfast
Lateral shoot: A side branch that comes off a primary shoot.
Intact shoot: A branch that has is able to grow from the tips of the branches out.
Print Data Sheets using the links below:
- Rockweed Phenophase Datasheet: Procedures A & B (PDF)
- Rockweed Age & Growth Data Sheet: Procedures B, C (PDF). Updated with datasheet for 2017 season).
Data Entry Links:
- Rockweed Phenophase Data Entry: Procedures A & B
- Rockweed Age and Growth Data Entry: Procedures B & C. Updated with link to 2017 data entry portal.





